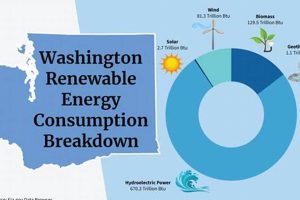
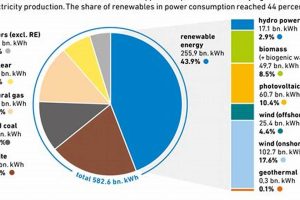
The intersection of chemical science and sustainable power generation is a critical area of research and development. For example, electrochemistry plays a crucial role in battery technologies for energy storage, while chemical catalysis drives the efficient production of fuels from renewable sources like sunlight and water. Materials science is another important component, as the development of novel materials with tailored properties is essential for improving the performance and durability of renewable energy technologies.
Developing efficient and cost-effective energy storage and conversion technologies is vital for transitioning to a sustainable energy future. This field offers solutions to the intermittency challenges posed by renewable sources like solar and wind power, paving the way for greater grid stability and reduced reliance on fossil fuels. Historically, advancements in chemical understanding have been central to energy innovation, from the development of more efficient combustion engines to the emergence of photovoltaics. Now, the focus has shifted to refining these processes and creating entirely new ones, tailored to harness renewable sources effectively.
This article will further explore specific areas where chemical sciences contribute significantly to renewable energy progress, including battery technology, solar fuels, hydrogen production, and materials development. The following sections will delve into the underlying chemical principles, current challenges, and future prospects of these vital technologies.
Tips for Advancing Sustainable Energy through Chemical Research
Progress in sustainable energy requires focused efforts across multiple disciplines. The following tips highlight key areas where advancements in chemical sciences can significantly contribute to a cleaner energy future.
Tip 1: Focus on Electrolyte Optimization: Advancements in electrolyte materials are crucial for improving battery performance, safety, and lifespan. Research efforts should concentrate on developing novel electrolytes with higher ionic conductivity, wider electrochemical windows, and enhanced thermal stability.
Tip 2: Explore Novel Catalyst Materials: Developing efficient and cost-effective catalysts is essential for various renewable energy processes, such as water splitting for hydrogen production and CO2 reduction to valuable fuels. Exploration of novel materials, including earth-abundant elements, is paramount.
Tip 3: Enhance Photovoltaic Efficiency: Improving the efficiency of solar cells through innovative materials and device architectures is crucial for maximizing solar energy conversion. Research should focus on developing new materials with enhanced light absorption and charge transport properties.
Tip 4: Develop Sustainable Energy Storage Solutions: Beyond traditional batteries, exploring alternative energy storage solutions like flow batteries and pumped hydro storage is critical for addressing the intermittency of renewable energy sources and ensuring grid stability.
Tip 5: Investigate Artificial Photosynthesis: Mimicking natural photosynthesis through artificial systems offers a promising pathway for converting sunlight into chemical fuels. Research should focus on developing efficient photocatalytic systems for water oxidation and CO2 reduction.
Tip 6: Promote Material Durability and Recyclability: Designing materials with enhanced durability and recyclability is essential for minimizing the environmental impact of renewable energy technologies. Research efforts should address the challenges of material degradation and develop efficient recycling processes.
By focusing on these key areas, research in chemical sciences can unlock significant advancements in renewable energy technologies, paving the way for a more sustainable future.
These recommendations offer a starting point for targeted research efforts. Continued exploration and innovation in these areas are vital for realizing the full potential of renewable energy and mitigating the effects of climate change.
1. Electrolysis
Electrolysis, a cornerstone of chemical science, plays a crucial role in renewable energy systems by enabling the production of hydrogen fuel and facilitating energy storage. Its ability to drive chemical reactions using electricity makes it a key technology for converting renewable energy sources into storable and transportable fuels, thus addressing the intermittency challenges associated with solar and wind power.
- Hydrogen Production
Electrolysis of water is the primary method for producing green hydrogen, a clean energy carrier. This process uses electricity, ideally from renewable sources, to split water molecules into hydrogen and oxygen. The hydrogen generated can be stored, transported, and used in fuel cells or combustion engines, offering a versatile and sustainable energy solution.
- Energy Storage
Electrolysis contributes to energy storage by enabling the production of hydrogen, which can be stored and later used to generate electricity through fuel cells. This process effectively stores the intermittent energy generated from renewable sources like solar and wind, ensuring a continuous power supply. Electrolysis also plays a role in other energy storage technologies, such as redox flow batteries.
- Chemical Synthesis
Beyond hydrogen production, electrolysis drives various chemical synthesis processes crucial for renewable energy. For instance, it can be used to produce ammonia, a key ingredient in fertilizers, using renewable energy instead of fossil fuels. Electrolysis can also facilitate the production of other valuable chemicals and fuels from renewable feedstocks.
- Grid Balancing
Electrolysis systems can respond rapidly to fluctuations in electricity supply and demand, making them valuable for grid balancing and stabilization. When excess renewable energy is available, it can be used for electrolysis, storing the energy as hydrogen. Conversely, during periods of high demand or low renewable energy generation, hydrogen can be converted back into electricity via fuel cells, ensuring grid stability.
These facets of electrolysis highlight its significance in the broader context of chemistry-driven renewable energy solutions. From fuel production and energy storage to chemical synthesis and grid balancing, electrolysis provides vital pathways for transitioning towards a sustainable energy future, reducing reliance on fossil fuels, and mitigating the impact of climate change.
2. Catalysis
Catalysis is essential for accelerating chemical reactions central to renewable energy technologies. It reduces the energy required for reactions to occur, thereby enhancing efficiency and enabling processes that would otherwise be impractical. The development and optimization of catalysts directly impact the viability and cost-effectiveness of renewable energy solutions. For example, efficient catalysts are critical for splitting water into hydrogen and oxygen using sunlight (photocatalytic water splitting), a key process for producing clean hydrogen fuel.
In fuel cells, catalysts facilitate the electrochemical reactions that convert chemical energy directly into electricity. Electrocatalysts enhance the oxygen reduction reaction (ORR) and hydrogen oxidation reaction (HOR) at the fuel cell electrodes, maximizing energy output. Furthermore, catalysis plays a crucial role in converting biomass into biofuels, and in the development of sustainable synthetic fuels. Developing robust and cost-effective catalysts for these processes is essential for diversifying energy sources and reducing reliance on fossil fuels.
Advancements in catalyst design, including the use of nanomaterials and single-atom catalysts, offer significant potential for improving the performance and durability of renewable energy technologies. However, challenges remain, such as developing catalysts from earth-abundant materials, enhancing catalyst stability under operating conditions, and understanding the detailed mechanisms of catalytic reactions to enable rational design and optimization. Overcoming these challenges will be crucial for unlocking the full potential of catalysis in driving the transition to a sustainable energy future.
3. Photovoltaics
Photovoltaics (PV), the direct conversion of sunlight into electricity, stands as a cornerstone of renewable energy technologies. The underlying principles of PV rely heavily on the chemical properties of semiconductor materials, highlighting the intrinsic connection between chemistry and sustainable energy generation. The efficiency, cost-effectiveness, and environmental impact of PV technologies are directly influenced by the chemical composition, structure, and processing of these materials.
- Semiconductor Materials
Semiconductors, materials with electrical conductivity between that of conductors and insulators, are the heart of photovoltaic devices. Silicon, the most widely used PV material, exemplifies the role of chemical purity and crystalline structure in determining solar cell efficiency. Other semiconductor materials, such as cadmium telluride (CdTe) and copper indium gallium selenide (CIGS), offer alternative approaches with distinct chemical properties and performance characteristics.
- Doping and Junction Formation
The controlled introduction of impurities (doping) into semiconductor materials alters their electronic properties, creating p-n junctions crucial for generating a voltage under illumination. The specific chemical dopants used and the precise control of their concentration are critical for optimizing the performance of solar cells. This process exemplifies the delicate interplay of chemistry and materials science in PV technology.
- Light Absorption and Charge Separation
The chemical composition and structure of the semiconductor material dictate its light absorption properties. Efficient PV materials maximize the absorption of photons from the solar spectrum, generating electron-hole pairs. The subsequent separation and collection of these charges at the electrodes are crucial for generating electrical current. Understanding and manipulating these chemical processes are key to improving solar cell efficiency.
- Stability and Degradation
The long-term performance of PV modules is influenced by the chemical stability of the constituent materials. Factors such as exposure to moisture, oxygen, and ultraviolet radiation can lead to chemical degradation, affecting the efficiency and lifespan of solar cells. Developing chemically robust materials and protective coatings is essential for ensuring the durability and sustainability of PV technologies.
These facets underscore the pivotal role of chemical research and development in advancing photovoltaic technologies. From the fundamental properties of semiconductor materials to the intricate processes of doping, light absorption, and degradation, chemistry provides the foundation for improving the efficiency, cost-effectiveness, and long-term sustainability of solar energy conversion, contributing significantly to the global transition towards cleaner energy sources.
4. Energy Storage
Energy storage is inextricably linked to the advancement of renewable energy technologies. The intermittent nature of renewable sources, such as solar and wind, necessitates efficient and reliable methods for storing the generated energy for use when demand exceeds production or when the source is unavailable. Chemistry plays a pivotal role in developing and optimizing various energy storage solutions, addressing the challenge of intermittency and enabling the wider adoption of renewable energy sources.
- Batteries
Batteries represent a prominent energy storage technology reliant on electrochemical principles. Chemical reactions within the battery convert stored chemical energy into electrical energy. Lithium-ion batteries, for instance, utilize the movement of lithium ions between electrodes to store and release energy. Research in battery chemistry focuses on improving energy density, charging rates, lifespan, and safety, as well as exploring alternative battery chemistries based on more abundant and sustainable materials.
- Pumped Hydro Storage
While not directly reliant on chemical reactions in the same way as batteries, pumped hydro storage benefits from advancements in materials science, a subfield of chemistry. Durable and corrosion-resistant materials are essential for the construction of reservoirs, dams, and turbines used in pumped hydro systems. Research in materials science contributes to the longevity and efficiency of this storage method, allowing for effective large-scale energy storage based on the potential energy of water.
- Flow Batteries
Flow batteries store energy in liquid electrolytes containing redox-active species. The chemical composition of these electrolytes and the reactions occurring at the electrodes determine the battery’s performance. Research in flow battery chemistry focuses on developing electrolytes with high energy density, long cycle life, and low cost, enabling grid-scale energy storage to support the integration of renewable energy sources.
- Thermal Energy Storage
Thermal energy storage utilizes materials with high heat capacity to store thermal energy generated from renewable sources or industrial processes. The chemical properties of these materials, such as their specific heat and thermal conductivity, determine the effectiveness of the storage system. Molten salt, for example, is used in concentrated solar power plants to store thermal energy for electricity generation when sunlight is unavailable. Research in this area focuses on developing materials with enhanced thermal properties and stability for efficient and cost-effective thermal energy storage.
The advancements in these energy storage technologies, driven by chemical research and development, are crucial for realizing the full potential of renewable energy. By addressing the inherent intermittency of renewable sources, energy storage solutions facilitate a stable and reliable energy supply, enabling the transition towards a more sustainable energy future and reducing reliance on fossil fuels.
5. Biofuels
Biofuels represent a class of renewable fuels derived from biomass, offering a sustainable alternative to fossil fuels. The production and utilization of biofuels are intrinsically linked to chemical processes, highlighting the crucial role of chemistry in renewable energy. From the conversion of biomass to usable fuels to the optimization of combustion and emission characteristics, chemical principles underpin the development and advancement of biofuel technologies.
- Biomass Conversion
Transforming biomass into usable fuels relies heavily on chemical processes. Methods such as transesterification convert plant oils and animal fats into biodiesel, while fermentation converts sugars into bioethanol. These processes involve intricate chemical reactions requiring careful control of parameters like temperature, pressure, and catalysts. Research in this area focuses on developing more efficient and cost-effective conversion methods to maximize biofuel yields and minimize environmental impact.
- Fuel Properties and Combustion
The chemical composition of biofuels directly influences their combustion characteristics, including energy content, ignition temperature, and emissions profile. Understanding and optimizing these properties are crucial for ensuring compatibility with existing engines and minimizing the environmental impact of biofuel combustion. Research explores the chemical modifications of biofuels to enhance their performance and reduce emissions of pollutants such as particulate matter and nitrogen oxides.
- Feedstock Development and Sustainability
The sustainable production of biofuels necessitates careful consideration of feedstock sources. Utilizing dedicated energy crops can compete with food production and impact land use. Research focuses on exploring alternative feedstocks, such as agricultural residues, algae, and dedicated non-food crops, and developing efficient methods for converting these diverse biomass sources into usable fuels. Chemical analysis of feedstocks is crucial for optimizing conversion processes and maximizing biofuel yields.
- Environmental Impact and Life Cycle Assessment
Assessing the overall environmental impact of biofuels requires a comprehensive understanding of the entire life cycle, from feedstock production to fuel consumption and waste management. Chemical analysis plays a crucial role in quantifying greenhouse gas emissions, water consumption, and other environmental impacts associated with biofuel production and use. Life cycle assessments provide valuable insights for optimizing biofuel production pathways and minimizing their environmental footprint.
These facets highlight the integral role of chemistry in advancing biofuel technologies. From the fundamental chemical processes involved in biomass conversion to the analysis of fuel properties, feedstock development, and environmental impact, chemistry provides the scientific foundation for developing sustainable and efficient biofuels. Continued research and development in biofuel chemistry are crucial for diversifying energy sources, reducing reliance on fossil fuels, and mitigating the effects of climate change.
Frequently Asked Questions about the Role of Chemistry in Renewable Energy
This section addresses common inquiries regarding the intersection of chemistry and renewable energy, providing concise and informative responses.
Question 1: How does chemistry contribute to improving the efficiency of solar cells?
Chemical research plays a crucial role in developing new materials for solar cells that absorb a broader range of wavelengths from the solar spectrum. Additionally, chemical processes are essential for optimizing the structure and composition of solar cells to enhance charge separation and transport, ultimately increasing their efficiency.
Question 2: What is the role of catalysis in renewable energy technologies?
Catalysis accelerates chemical reactions crucial for renewable energy processes, such as water splitting for hydrogen production, fuel cell operation, and biofuel synthesis. Efficient catalysts reduce the energy required for these reactions, improving overall efficiency and cost-effectiveness.
Question 3: How does chemistry address the intermittency of renewable energy sources?
Chemical research is central to developing advanced energy storage solutions, such as batteries and flow batteries. These technologies rely on chemical reactions to store and release energy, providing a stable power supply even when renewable sources are unavailable.
Question 4: What are the environmental benefits of biofuels, and how does chemistry contribute to their development?
Biofuels offer the potential to reduce greenhouse gas emissions compared to fossil fuels. Chemistry plays a critical role in developing efficient and sustainable methods for converting biomass into biofuels, optimizing fuel properties, and assessing their environmental impact.
Question 5: How does chemical research contribute to the development of more sustainable energy storage materials?
Research focuses on developing battery chemistries based on earth-abundant and less toxic materials. Chemical processes are also essential for improving the recyclability of battery components, minimizing the environmental impact of energy storage technologies.
Question 6: What are some of the key challenges in applying chemistry to renewable energy technologies?
Challenges include developing cost-effective and durable materials, improving the efficiency of catalytic processes, and scaling up laboratory-scale advancements to industrial production levels. Addressing these challenges requires ongoing research and innovation in chemical science.
Understanding the chemical principles behind renewable energy technologies is essential for developing innovative solutions for a sustainable energy future. Continued research and development in this field are crucial for addressing the global energy challenge and mitigating climate change.
For further exploration, the following section delves deeper into specific renewable energy technologies and their chemical underpinnings.
The Indispensable Role of Chemistry in Renewable Energy
This exploration has underscored the profound impact of chemical sciences on renewable energy technologies. From the electrochemical reactions driving batteries and fuel cells to the catalytic processes optimizing biofuel production and the material science advancements enhancing solar cell efficiency, chemistry is fundamental to the transition toward sustainable energy. The development of novel materials, the optimization of chemical processes, and the fundamental understanding of energy conversion and storage mechanisms are all critical areas where chemical research drives innovation and progress in the renewable energy sector. This interdisciplinary field addresses key challenges, including intermittency, energy density, and cost-effectiveness, paving the way for a future powered by clean and sustainable resources.
The future of sustainable energy hinges on continued advancements in chemical research. Investing in research and development, fostering collaboration across disciplines, and promoting the implementation of innovative chemical solutions are crucial steps towards a future powered by clean, reliable, and affordable renewable energy. The transition to a sustainable energy landscape requires a concerted global effort, with chemical sciences playing a central role in shaping a future where energy production and consumption are decoupled from environmental degradation.